Discussion
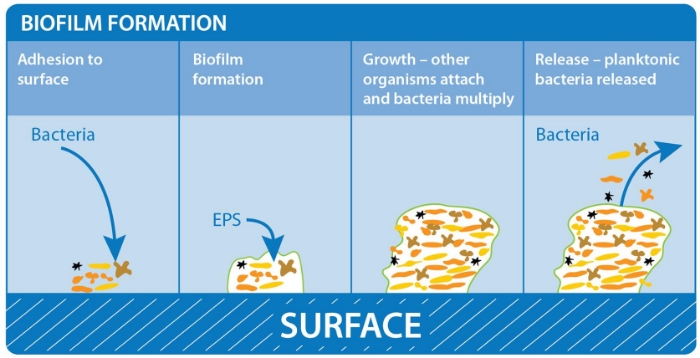
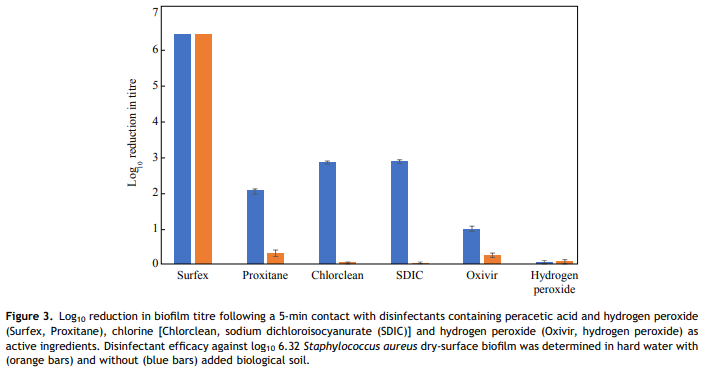
In this study, S. aureus DSB [16] was chosen for testing hospital surface disinfectants as 50% of clinical biofilms incorporate S. aureus [7] which commonly causes HAI [19]. The efficacy of three formulated disinfectants, based on three differing active ingredients (chlorine, hydrogen peroxide and peracetic acid), along with generic (unformulated) solutions containing these three active ingredients were evaluated. In this manner, the excipient (non-active) ingredients, as well as the active ingredients themselves, could be evaluated. Tests were undertaken in the presence of organic soil, as combined cleaning/disinfecting systems are becoming more popular as clinical surfaces are often not precleaned prior to disinfection. Thus, efficacy testing in the presence of large amounts of organic soil is more reflective of worse-case clinical conditions. This study evaluated three formulated, commercially available disinfectant systems, each of which contained an oxidizing biocide, along with other ingredients such as surfactants. The effect of the addition of the proprietary ingredients to disinfectant efficacy was evaluated by comparing the formulated disinfectants with generic equivalents in a bid to determine if biofilm removal is due to the active ingredient alone or if the proprietary ingredients act in synergy with the active ingredient. The outstanding performer in this study was Surfex, which completely inactivated the DSB in the presence or absence of soil. The formulated chlorine-based product Chlorclean, and unformulated SIDC tablets, were the next best performers, although they killed significantly fewer biofilm bacteria (3 log10) than Surfex (P < 0.001) and only in the absence of soil. Previous studies have demonstrated that chemicals such as hypochlorite are consumed by the surface layers of the biofilm neutralizing the disinfectant before it can penetrate into deeper layers [20], making hydrated biofilm more tolerant than planktonic cells to these disinfectants [12]. However, a study on the efficacy of hypochlorite against DSB found that this semi-dehydrated biofilm was more tolerant to hypochlorite than hydrated biofilm [11]. The water content of hydrated S. aureus biofilm grown in the CDC bioreactor is 90%, whilst that of DSB is 61% [21]. This lower water content, in combination with the thicker extracellular polymeric substances (EPS), may result in lower diffusion of biocides and hence contribute to biocide tolerance.
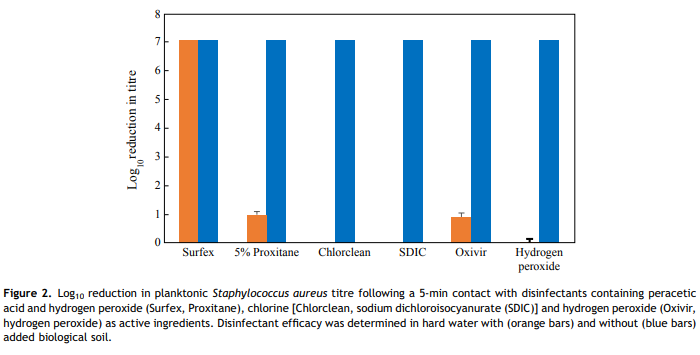
Even in the absence of soil, the hydrogen-peroxide-based disinfectants killed significantly less biofilm bacteria than disinfectants based on chlorine or a combination of peracetic acid and hydrogen peroxide (P < 0.001). Oxivir killed approximated 1 log10 of the biofilm bacteria, while hydrogen peroxide solution had no effect; however, the manufacturer-recommended contact time for Oxivir for killing bacteria is 10 min, not 5 min as used in the study, and this could explain its lower performance. However, even a contact time of 5 min is probably excessive given the way in which dry hospital surfaces are cleaned. The majority of disinfectants have no residual effect and are only active when wet.
The difference in kill rates between Surfex (formulated additives) and Proxitane (no additives) suggests that the activity of Surfex against DSB may be governed not only by the active ingredients (hydrogen peroxide and peracetic acid), but also by other factors such as the added surfactants or excipients, chelating agents or its solution pH. Surfactants may increase diffusion of the active ingredients into the biofilm (due to a lowering of the solution surface tension, and hence improved wetting of the biofilm surface). Increased diffusion is likely to result in increased biofilm kill as all of the tested disinfectants, in the absence of organic soil, can kill 7 log10 of planktonic organisms. Chelating agents complex any calcium and magnesium ions present in the hard water, plus any other interfering metals often present in tap water such as iron and manganese, and thus increase disinfectant performance in hard water. Additionally, the source of peracetic acid in the two disinfectants is different, which under certain circumstances (e.g. disruption of Proxitane equilibrium) may affect levels of active ingredients. Proxitane is an equilibrium mixture formed by the reaction between hydrogen peroxide and acetic acid according to the following formula: H2O2 þ CH3CO2H # CH3CO3H þ H2O [18]. However, in Surfex, the peracetic acid is generated by the reaction of hydrogen peroxide with tetraacetylethylenediamine [22]. The source of hydrogen peroxide in Surfex is sodium peroxycarbonate, a 2:3 complex of hydrogen peroxide and sodium carbonate, that releases hydrogen peroxide on dissolution in water.
Except for Surfex, the efficacy of disinfectants was significantly decreased by the addition of soil, with little or no reduction in the viable bacteria load. This result is in agreement with most reports of chlorine disinfectants, where serious loss of efficacy has been demonstrated by the presence of organic matter [23] and hard water [24,25]. Both hydrogen peroxide and peracetic acid are effective oxidizing biocides. This study showed that the addition of organic soil had no effect on the efficacy of Surfex, whilst the generic equivalent, diluted Proxitane, was inactivated. This is most likely due to the other ingredients within the formulation, such as chelating agents, or perhaps due to the differences in pH (8.10 for Surfex vs 2.6 for a 4% solution of Proxitane). Compared with hydrogen peroxide, peracetic acid has the disadvantage that it is less stable when diluted, dissociating into acetic acid and hydrogen peroxide over a matter of hours due to the shift in equilibrium conditions brought on by dilution in water.
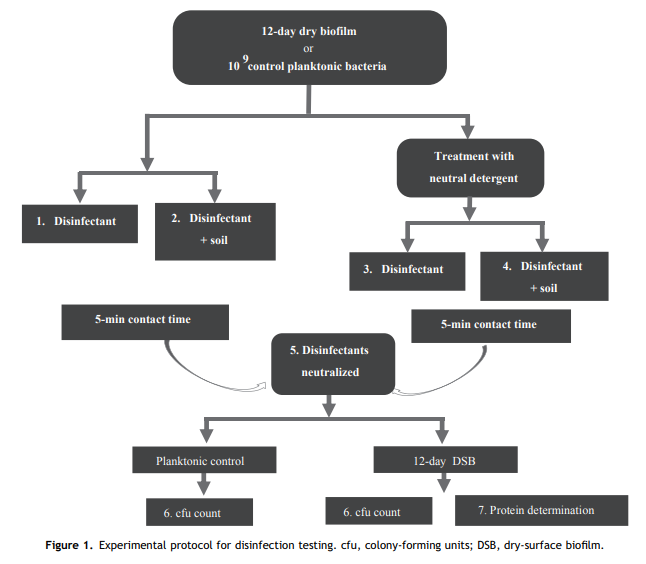
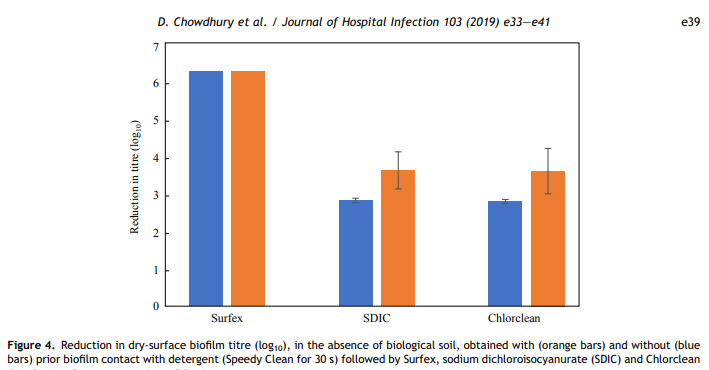
The very short detergent treatment used in this study was to simulate someone gently wiping over a surface with a damp cloth, thus wetting the surface of the DSB with surfactants to increase biocide activity. This detergent treatment had no significant effect on the efficacy of the three biocides tested (Chlorclean, SDIC and Surfex). However, even if hospital surfaces are precleaned, the likelihood of DSB being present is high [7e9].
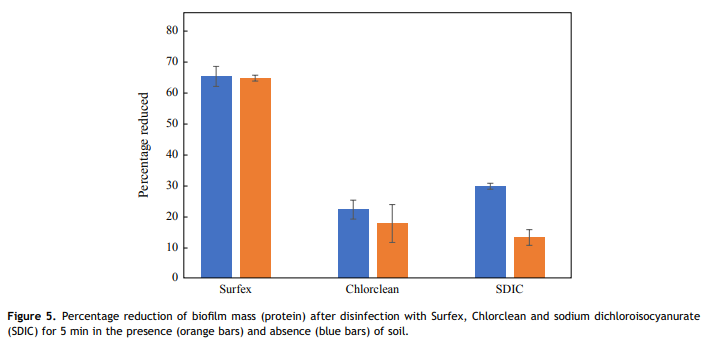
Almatroudi et al. [16] demonstrated that protein was a principal component (56%) of both the in-vitro DSB model and biofilms contaminating dry clinical surfaces in hospitals with protein contents varying from 42% to 95%. Therefore, the present study measured residual protein on the treated coupons to determine the proportion of biofilm mass removed by the oxidizing action of the disinfectants. None of the disinfectants were able to completely remove all biofilm protein with a 5-min contact time; however, a higher percentage reduction of biofilm protein was observed in 5 min with Surfex (65%) than the other tested disinfectants (<30%), both in the presence and absence of soil.><30%), both in the presence and absence of soil.
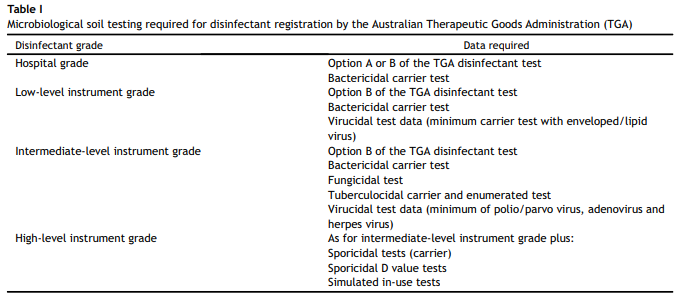
In conclusion, disinfectant efficacy against biofilm can vary significantly, despite containing similar levels of biocides, due to their formulation/additives. The disinfectant formulation also affects disinfectant action in the presence of soil. Therefore, it is crucial to select clinically efficient disinfectant agents with the potential of effectively eradicating dry biofilm from hospital environments. It is suggested that purchasing officers should ask disinfectant manufacturers for efficacy testing results, and consider whether efficacy testing has been conducted in the presence of biological soil and/or dry biofilm.
Conflict of interest statement
Whiteley Corporation was the industrial partner associated with the Australian Research Council Linkage Project. They are a manufacturer of disinfectants and detergents for use in health care and one of their products was tested in this study. Their role did not lead to any bias in formulating, executing, analysing or writing up the research.
Funding source
KV was in receipt of a Macquarie University Vice Chancellor Innovation Fellowship. This work was funded, in part, by the Faculty of Medicine and Health Sciences, Macquarie University and by an Australian Research Council (ARC) Linkage Grant LP130100572, ‘Developing novel chemistries for removing environmental surface biofilms to reduce hospital acquired infections’. An ARC Linkage grant requires an industry partner that is required by law to provide ‘in kind’ materials to the university. The funders contributed to the study design, provided the disinfectants as part of their obligation under the rules of the linkage grant, and supplied patent information. The funders had no role in data collection and analysis, decision to publish, or preparation of the manuscript.
References
[1] Ling ML, Apisarnthanarak A, Madriaga G. The burden of healthcare-associated infections in southeast Asia: a systematic literature review and meta-analysis. Clin Infect Dis 2015;60:1690e9.
[2] Weinstein RA. Epidemiology and control of nosocomial infections in adult intensive care units. Am J Med 1991;91:179e82.
[3] Hayden MK, Blom DW, Lyle EA, Moore CG, Weinstein RA. Risk of hand or glove contamination after contact with patients colonized with vancomycin-resistant enterococcus or the colonized patients’ environment. Infect Control Hosp Epidemiol 2008;29:149e54.
[4] Carling PC, Bartley JM. Evaluating hygienic cleaning in health care settings: what you do not know can harm your patients. Am J Infect Control 2010;38:S41e50.
[5] Dancer SJ. Controlling hospital-acquired infection: focus on the role of the environment and new technologies for decontamination. Clin Microbiol Rev 2014;27:665e90.
[6] Mitchell BG, Dancer SJ, Anderson M, Dehn E. Risk of organism acquisition from prior room occupants: a systematic review and meta-analysis. J Hosp Infect 2015;91:211e7.
[7] Hu H, Johani K, Gosbell IB, Jacombs A, Almatroudi A, Whiteley GS, et al. Intensive care unit environmental surfaces are contaminated by multiresistant bacteria in biofilms: combined results of conventional culture, pyrosequencing, scanning electron microscopy and confocal laser microscopy. J Hosp Infect 2015;91:35e44.
[8] Johani K, Abualsaud D, Costa DM, Hu H, Whiteley G, Deva A, et al. Characterization of microbial community composition, antimicrobial resistance and biofilm on intensive care surfaces. J Infect Public Health 2017;11:418e24.
[9] Ledwoch KD, Dancer SJ, Otter JA, Kerr K, Roposte D, Maillard J-Y. Beware biofilm! Dry biofilms containing bacterial pathogens on multiple healthcare surfaces; a multi-centre study. J Hosp Infect 2018;100:e47e56.
[10] Hadi R, Vickery K, Deva A, Charlton T. Biofilm removal by medical device cleaners: comparison of two bioreactor detection assays. J Hosp Infect 2010;74:160e7.
[11] Almatroudi A, Gosbell IB, Hu H, Jensen SO, Espedido BA, Tahir S, et al. Staphylococcus aureus dry-surface biofilms are not killed by sodium hypochlorite: implications for infection control. J Hosp Infect 2016;93:263e70.
[12] Otter JA, Vickery K, Walker JT, deLancey Pulcini E, Stoodley P, Goldenberg SD, et al. Surface-attached cells, biofilms and biocide susceptibility: implications for hospital cleaning and disinfection. J Hosp Infect 2015;89:16e27.
[13] AS/NZS. AS/NZS 4187:2014 Reprocessing of reusable medical devices in health service organizations. Sydney: Standards Australia Limited and Standards New Zealand; 2014.
[14] TGA.Therapeutic Goods Order No. 54. Standards for disinfectants and sterilants. Canberra: Australian Government; 2009.
[15] ISO14937:2009. Sterilization of health care products e general requirements for characterization of sterilizing agent and the development, validation and routine control of a sterilization process for medical devices. Geneva: International Organization for Standardization; 2009.
[16] Almatroudi A, Hu H, Deva A, Gosbell IB, Jacombs A, Jensen SO, et al. A new dry-surface biofilm model: an essential tool for efficacy testing of hospital surface decontamination procedures. J Microbiol Methods 2015;117:171e6.
[17] Sanosil Disinfectants for Life. The safety data sheet. West Perth: Risk Management Technologies; 2015. Available at: http://www.helixsolutions.net.au/sites/helixsolutionsnetau/assets/public/ Image/PDFs/Chlor-Clean_Tablets_H8950_SDS.pdf [last accessed November 2018].
[18] Ramirez JA, Omidbakhsh N. Patent Cooperation Treaty Application No. WO03067989. Munich: European Patent Office; 2003.
[19] Tong SYC, Davis JS, Eichenberger E, Holland TL, Fowler VG. Staphylococcus aureus infections: epidemiology, pathophysiology, clinical manifestations, and management. Clin Microbiol Rev 2015;28:603e61.
[20] Chen X, Stewart PS. Chlorine penetration into artificial biofilm is limited by a reaction-diffusion interaction. Environ Sci Technol 1996;30:2078e83.
[21] Almatroudi A, Tahir S, Hu H, Chowdhury D, Gosbell IB, Jensen SO, et al. Staphylococcus aureus dry-surface biofilms are more resistant to heat treatment than traditional hydrated biofilms. J Hosp Infect 2018;98:161e7.
[22] Glasbey T. Patent Cooperation Treaty Application No. WO2015066760. Munich: European Patent Office; 2015.
[23] Lambert RJW, Johnston MD. The effect of interfering substances on the disinfection process: a mathematical model. J Appl Microbiol 2001;91:548e55.
[24] Davis B. Surfactantebiocide interactions. Recent developments in the technology of surfactants. Crit Rep Appl Chem 1990;30: 65e131.
[25] Holah J. Progress report on CEN/TC 216/Working Group 3: Disinfectant test methods for food hygiene, institutional, industrial and domestic applications. Int Biodeterior Biodegrad 1995;36:355e65.